 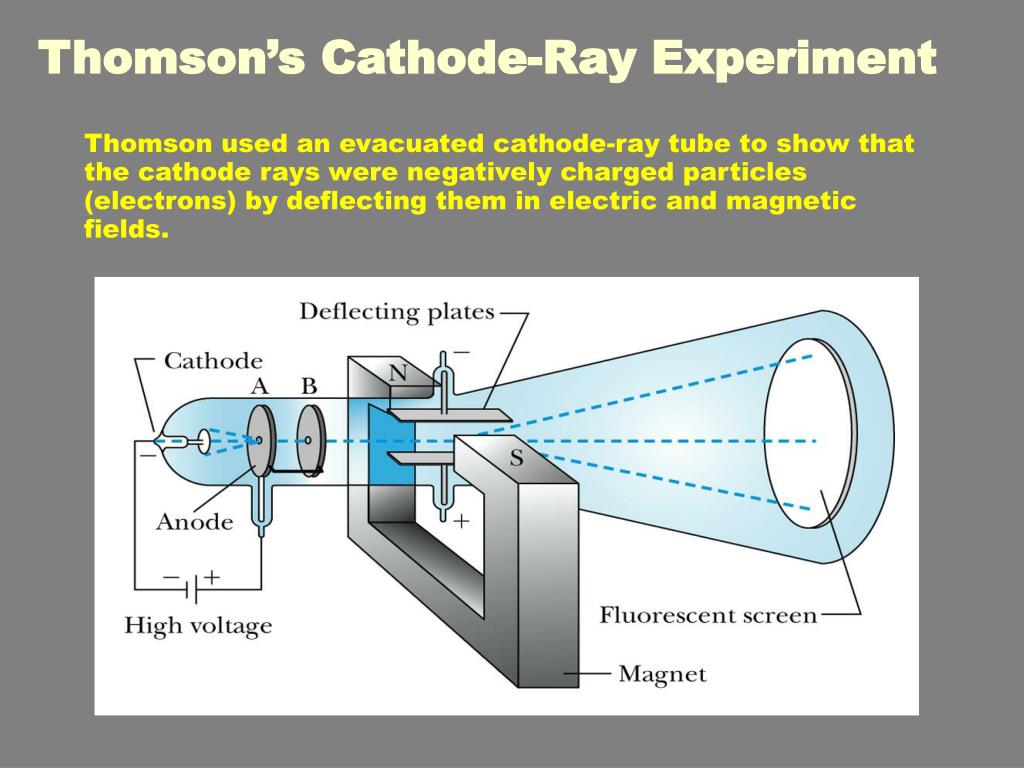
What evidence from the Cathode Ray Tube Experiment support the claim that electrons have a negative charge? The Cathode Ray bent way from a negatively charged plate. What evidence from the cathode ray tube experiment support the claim that electrons have a negative charge?Įlectrons can be deflected by a magnetic field. Using this information Thomson determined the mass to charge ratio of an electron. Then Thomson measured how much various strengths of magnetic fields bent the particles. Thomson was able to deflect the cathode ray towards a positively charged plate deduce that the particles in the beam were negatively charged. How did JJ Thomson determine the charge of an electron? Thomson’s cathode ray tube experiments provided the first evidence that atoms were composed of even smaller particles called electrons. What did JJ Thomson’s cathode ray experiments determine quizlet? As the electron is repelled by the nucleus of an atom(which is positively charged due to protons). An experiment called “oil drop experiment” determined charge on electron. How do we know that electrons are negative?Īs to electrons, they were detected by JJ Thomson. Thus it can be proved that electrons are negatively charged. Cathode rays also neutralise the gold leaf electroscope, charged with positive electricity. Under the influence of an electric field the cathode rays as well as the electron beam, are deflected towards the positive plate of the electric field. How do you prove an electron is negative? He deduced that the cathode rays were made up of negatively-charged particles. In the second experiment, he discovered that the charge in the cathode rays was negative. In Thomson’s first experiment, he discovered that cathode rays and the charge they deposited were intrinsically linked together. 
What experiment did he do explain what his experiment did and what it proved? … For example, electrons have negative charge and protons have positive charge, but neutrons have zero charge. 
Is there a negative electrical charge?Įlectric charges are of two general types: positive and negative. Positively charged ions are called cations, negatively charged ions, anions. What is known as a negatively charged ion? Bohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element. The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. Ernest Rutherford discovered the nucleus of the atom in 1911. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” What did Rutherford discover? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. He observed that cathode rays were deflected by a magnetic field in the same manner as a wire carrying an electric current, which was known to be negatively charged. 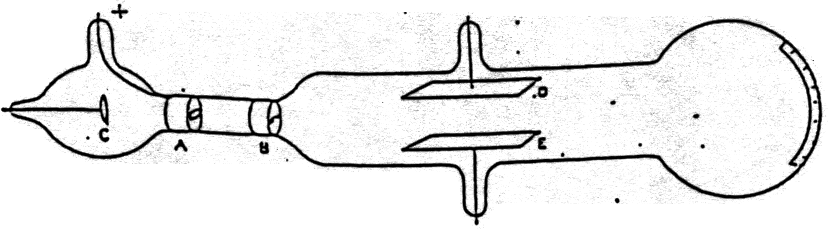
In order to determine if the cathode ray consisted of charged particles, Thomson used magnets and charged plates to deflect the cathode ray. How did Thomson determine that the electrons have a negative charge? Millikan discovered that there is a fundamental electric charge-the charge of an electron. Thomson’s cathode ray tube showed that atoms contain small, negatively charged particles called electrons. How were researchers able to determine that the electron has a negative charge? It was performed originally in 1909 by the American physicist Robert A. Millikan oil-drop experiment, first direct and compelling measurement of the electric charge of a single electron. What experiment did he do to understand the charge of electrons? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
